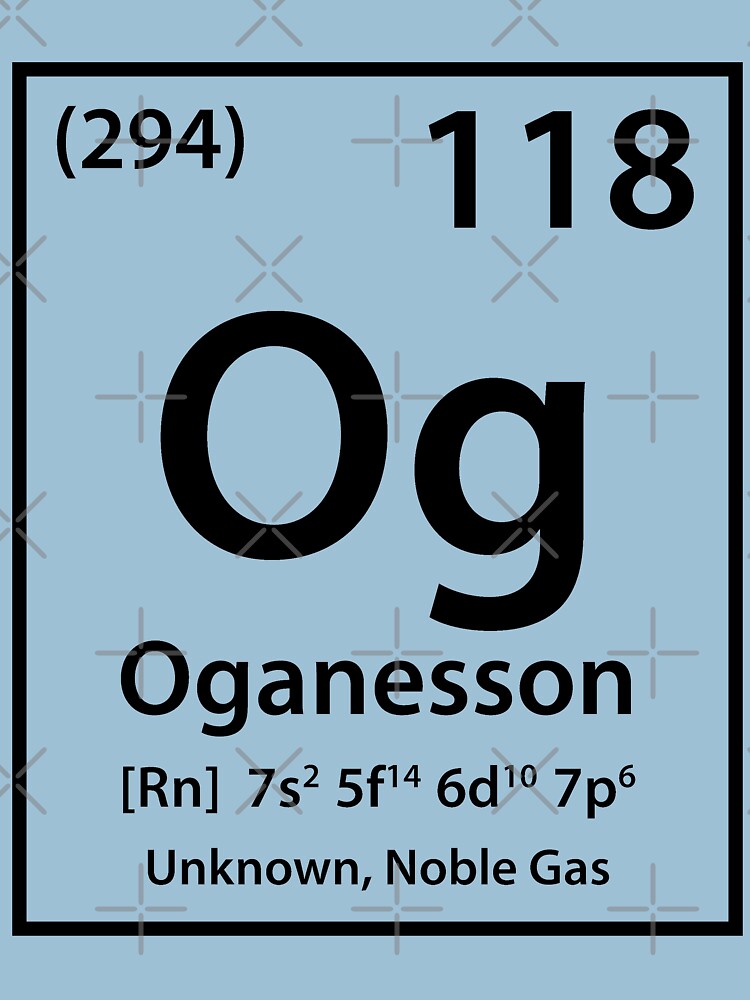
Professor Yuri Oganessian (born 1933) is a Russian nuclear physicist who is credited with three confirmed element discoveries.įirstly, of the 118 known elements, 94 were found naturally on Earth. Oganesson – Oganesson attributed its name to a scientist following the IUPAC naming convention.Tennessine – Tennessine recognises the laboratories that contribute to element research in the Tennessee region which include Oak Ridge National Laboratory, Vanderbilt University, and the University of Tennessee in Knoxville.Moscovium – Moscovium also represents a geographical region, specifically, the Moscow region which is where the discovery experiments were conducted.Nihon means “the Land of Rising Sun” and provides a direct connection to Japan as a nation. As this was the first element discovered by an Asian country, Japan wanted the name to represent the geographical region. Nihonium – Nihon is one way to say “Japan” in Japanese.a mythological concept or character (including an astronomical object),.Whilst these teams have the rights to name the elements, they do have to follow a naming convention as outlined by the IUPAC which states that any new element must be named after either: The US, Russia, and Japan were credited with the discovery of the new elements and the names were announced in June. The rights to name the elements were given to those research teams that discovered them. In January, the new elements were first announced and given interim names until formal names were released. Oganesson (symbol Og) – atomic number 118.Tennessine (symbol Ts) – atomic number 117.Moscovium (symbol Mc) – atomic number 115.Nihonium (symbol Nh) – atomic number 113.The new elements will be added to the lower right-hand corner and consist of: After the review period, the IUPAC Council will formally approve the revised periodic table. The International Union of Pure and Applied Chemistry (IUPAC) has worked with those laboratories that discovered the elements to name them and has now released the new periodic table for a five-month public review period which expires on the 8th of November, 2016. Polonium, for example, is dangerous due to its toxicity and radioactivity.In June 2016, it was publicly announced that the periodic table will have four new elements added to it. Some are found in drugs, cleaners, and pesticides. Metalloids are extremely important in electronics as semiconductors, although they are also used in optical fibers, alloys, glass, and enamels.As far as appearances go, metalloids range from dull to shiny.Oxidation numbers of elements in this group range from +3 to -2. Metalloids may either gain or lose electrons during chemical reactions.One exception is silicon, which is brittle. Semimetals are typically malleable and ductile.Semimetals/metalloids have high lattice dielectric constants and high diamagnetic susceptibilities.These elements have lower electrical and thermal conductivity than metals. 
Exceptions are silicon and germanium, which are true semiconductors, as they can conduct electricity under the right conditions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
